 
The non-integral, 3.6-residue-per-turn repeat of the alpha helix means that the Cα's of successive turns are about halfway offset, giving the main chain a distinctive 7-pointed star appearance in end view. The O and N atoms of the helix main chain are shown as red and blue balls, respectively. The kinemage linked above shows an individual alpha helix, viewed from the N-terminal end to resemble the "helical wheel" (see figure below). If you need information on using King, please hover here.Īn alpha helix is an element of secondary structure in which the amino acid chain is arranged in a spiral. Upon loading this page, the KiNG Java Applet should automatically spawn. To load the KiNG Java Applet, just click here. These will be described in the following sections and visualized using the KiNG software mentioned previously. Several common secondary structures have been identified in proteins. While primary structure describes the sequence of amino acids forming a peptide chain, secondary structure refers to the local arrangement of the chain in space. Each amino acid is connected to the next by a peptide bond. By convention, the primary structure of a protein is read and written from the amino-terminal (N) to the carboxyl-terminal (C) end. The primary structure of a peptide or protein is the linear sequence of its amino acids (AAs). Richardson (1992) Protein Science 1: 3-9. Reference: "THE KINEMAGE: A TOOL FOR SCIENTIFIC COMMUNICATION" D.C. Kinemages were originally implemented under the auspices of the Innovative Technology Fund and the Protein Society, and the programming and maintenance are done by David C. These "kinemages" (kinetic images) can be rotated, moved, and zoomed, and parts can be hidden or shown. This module includes links to KiNG (Kinemage, Next Generation), which displays three-dimensional structures in an animated, interactive format. As you traverse though this module please note these aspects. We also view structures in lots of ways - Cα backbone, ball-and-stick, CPK, ribbon, spacefilling - as well color is used to highlight different aspects of the amino acids, structure, etc. In this series of pages we examine the different levels of protein organization. Till next time.A 2014 Foundations of Medicine eLAB Levels of Protein OrganizationĪ protein's primary structure is defined as the amino acid sequence of its polypeptide chain secondary structure is the local spatial arrangement of a polypeptide's backbone (main chain) atoms tertiary structure refers to the three-dimensional structure of an entire polypeptide chain and quaternary structure is the three-dimensional arrangement of the subunits in a multisubunit protein. 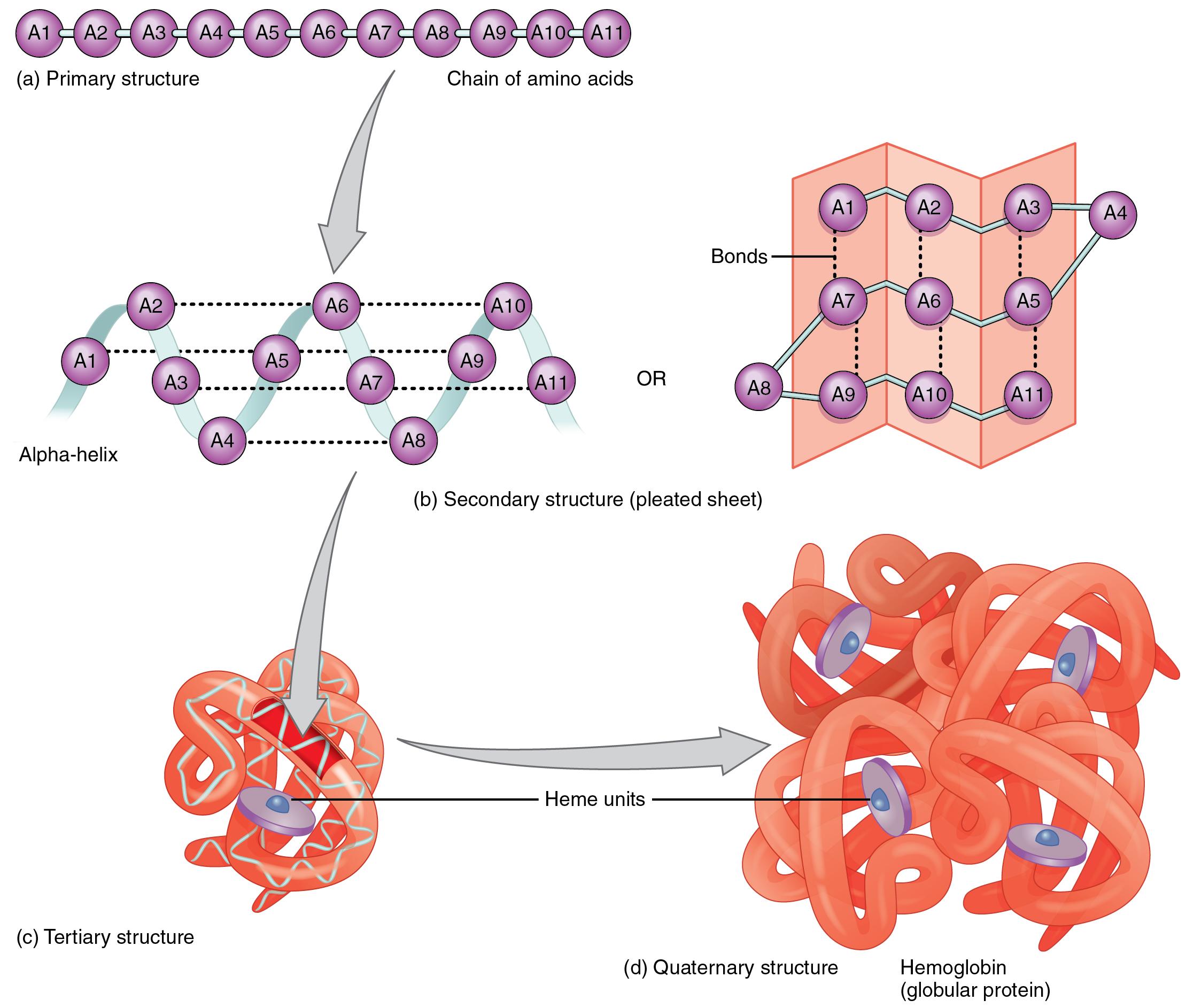 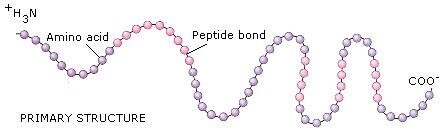
Ok guys hope this little information helps you somehow. This is why when women apply heat to their hair they can either straighten it or curl it. Of the four hydrogen bond which when heated the bonds breaks and reforms back into a formation. The diagram on the right hand side is showing the interactions between polypeptide bonds. Here more than one polypeptide chain interacts to form one unit which increases the function of the protein in the example of hemoglobin in blood which allows for the maximum transport of oxygen throughout the body in the red blood cells. There are four major interactions such as hydrogen bonding, ionic bonding, Disulphide Bridge and hydrophobic interaction. Proteins which contain more than one polypeptide chain are described as quaternary structures. When stability increases the hydrophobic effect is present, therefore allows for R group interactions. In a tertiary structure the amino acids in the linear system is arranged spatially and the residues that are located adjacent to the sequence is also of spatial arrangement. Each Hydrogen bond that occurs in the alpha helix is parallel, which can be seen in the picture below and at every turn of the alpha helix there are 3.6 residues present. The main type of bonding seen in the alpha helix is Hydrogen bonding, which takes place between a peptide bond. These properties are all found due to the make-up of their R groups.Ī secondary structurehas continuous regions of the polypeptide chains that folds in an alpha helix or beta pleated sheets. Some amino acids are hydrophobic (otherwise known as water hating), polar meaning they contain a hydroxyl group that is able to react with water and charged due to the negative or positive charge on them. Now as primary structure is the basic lineage of amino acids in a protein, note that every amino acid has its own properties. Today we will be looking at the numerous structures of proteins.Ī primary structure protein is composed of a liner system of amino acids that are linked together by polypeptide bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
